Introduction
When most practitioners first learn about multiple sclerosis disease, the explanation appears straightforward. The immune system mistakenly attacks the myelin sheath that protects nerve fibers within the central nervous system. Demyelination follows, neural signaling becomes disrupted, and neurological symptoms gradually appear.
This framework has dominated medical education for decades. Yet if you spend enough time working with individuals experiencing MS disease, you begin to notice something interesting. The autoimmune explanation describes what happens, but it rarely explains why it happens in the first place.
Patients often report years of fatigue, digestive disturbances, metabolic instability, chronic stress, and unexplained inflammation long before neurological symptoms begin. These patterns suggest that multiple sclerosis may represent a systems level breakdown rather than an isolated immune error.
For practitioners committed to understanding root cause medicine, this distinction matters greatly. If MS is viewed only as an autoimmune attack, treatment focuses almost entirely on immune suppression. But if MS develops from broader disturbances involving metabolism, gut health, mitochondrial function, and nervous system regulation, the clinical conversation becomes much richer.
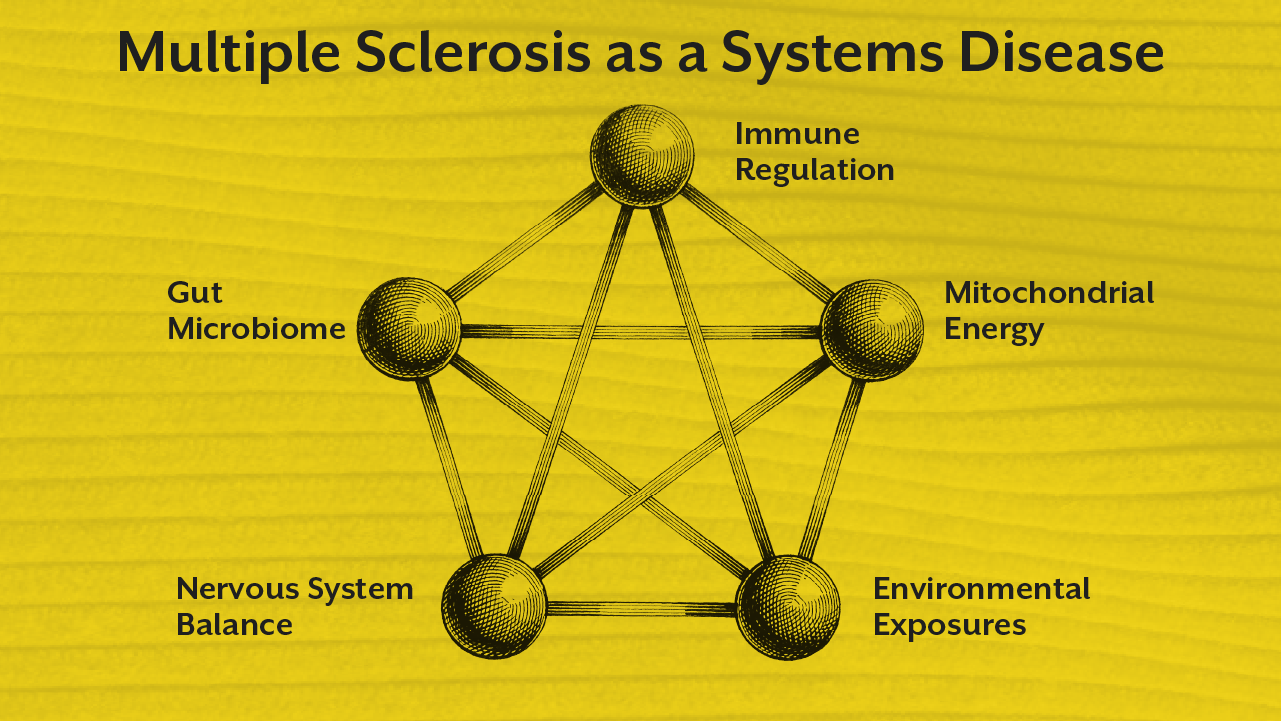
In functional and integrative medicine frameworks, MS disorder is increasingly understood as a complex interaction between immune signaling, cellular energy production, microbiome dynamics, and neuroinflammatory pathways.
This perspective does not reject autoimmunity. Instead, it asks a deeper question.
What conditions allow the immune system to attack the nervous system in the first place?
The Traditional Autoimmune Model of MS
Multiple sclerosis symptoms occur when immune cells cross the blood brain barrier and begin damaging the myelin sheath surrounding nerve fibers. Myelin acts as an electrical insulator, allowing rapid communication between neurons.
Once this protective coating is damaged, nerve impulses slow down or become disrupted entirely. This explains many of the classical neurological symptoms associated with ms multiple sclerosis, including muscle weakness, coordination issues, visual disturbances, and sensory abnormalities.
However, the autoimmune explanation does not fully explain several clinical realities observed in practice.
First, many individuals experience systemic symptoms years before neurological damage appears. Fatigue, metabolic instability, digestive dysfunction, and chronic inflammation are commonly reported in early stages of the ms medical condition.
Second, immune dysregulation rarely emerges spontaneously. Immune signaling is deeply connected to environmental exposures, microbiome balance, mitochondrial function, and metabolic stress.
In other words, the immune system is not acting alone. It is responding to signals from the broader physiological environment.
This is precisely where functional medicine perspectives begin to expand the conversation.
Interestingly, a similar systems biology perspective is discussed in the iThrive Academy article “5 Pillars of Gut Brain Health: From Inflammation to Neurotransmitters”, which explains how immune activity, microbiome balance, and nervous system signaling are deeply interconnected within the gut brain axis.
Understanding these connections helps practitioners move beyond a narrow autoimmune model toward a more integrated view of disease development.
The Gut Brain Axis in MS Development
One of the most compelling areas of research in causes of ms involves the relationship between gut health and neuroinflammation.
The gastrointestinal tract houses a vast ecosystem of microorganisms that continuously interact with the immune system. These microbes produce metabolites, regulate inflammatory pathways, and influence neurotransmitter synthesis.
When microbial balance becomes disrupted, a state known as dysbiosis can emerge. Dysbiosis alters immune signaling, increases intestinal permeability, and allows inflammatory molecules to enter systemic circulation.
Over time, this inflammatory burden may influence the central nervous system through several mechanisms.
First, circulating cytokines can weaken the blood brain barrier, allowing immune cells to access neural tissue. Second, microbial metabolites can influence microglial activity within the brain, increasing neuroinflammatory responses. Third, vagus nerve signaling may transmit inflammatory signals directly from the gut to the brain.
From a practitioner perspective, this explains why digestive disturbances frequently appear in individuals with multiple sclerosis symptoms long before neurological damage is diagnosed.
This relationship highlights an important clinical principle. Neurological disorders rarely originate exclusively in the brain. Instead, they often emerge from systemic regulatory imbalances affecting multiple organ systems simultaneously.

Cellular Energy and the Hidden Role of Mitochondria
Another overlooked factor in ms disorder involves mitochondrial function.
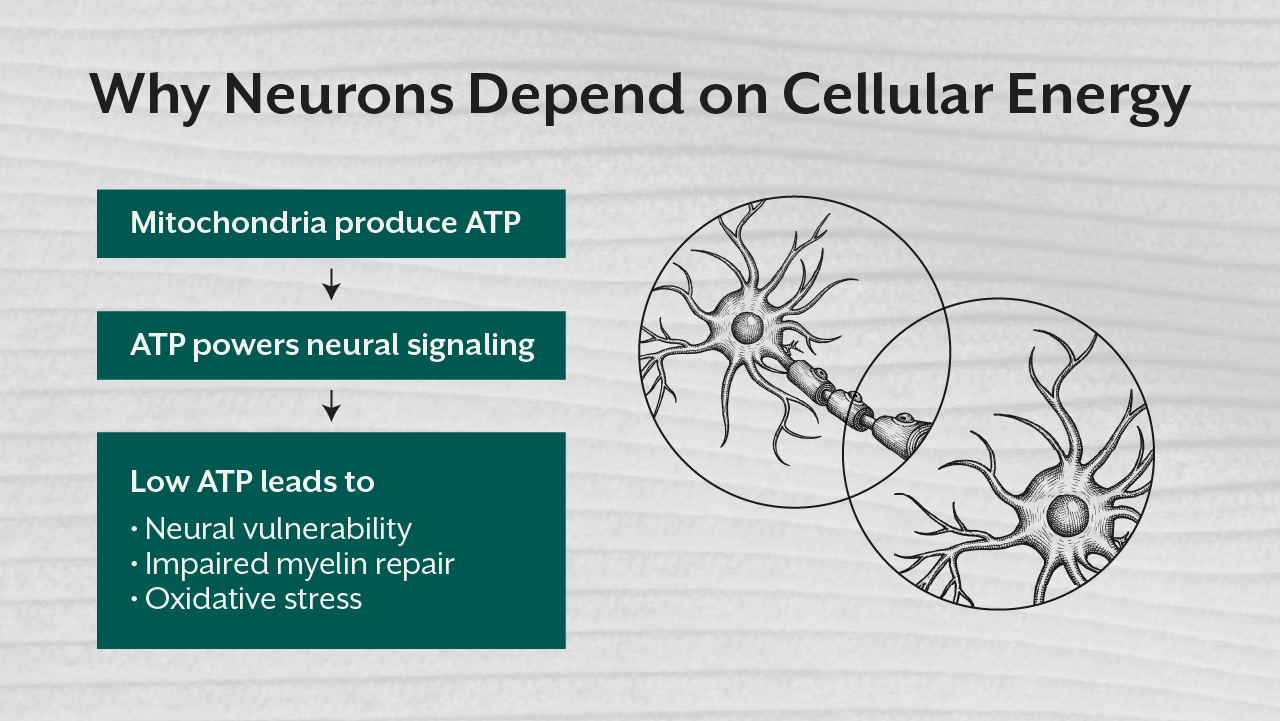
Neurons are among the most energy demanding cells in the human body. Maintaining electrical gradients, synthesizing neurotransmitters, and repairing damaged tissue requires continuous ATP production.
When mitochondrial efficiency declines, neural tissues become highly vulnerable to damage.
Several studies suggest that individuals with multiple sclerosis disease show signs of impaired mitochondrial activity, including reduced ATP production, increased oxidative stress, and altered metabolic signaling within neural cells.
This creates a dangerous biological environment.
Without sufficient cellular energy, neurons struggle to maintain structural integrity. Myelin repair slows down. Oxidative stress increases. Inflammatory pathways become amplified.
In this sense, mitochondrial dysfunction may not simply be a consequence of MS. It may contribute to the conditions that allow the disease to progress.
Practitioners studying chronic illness often recognize this pattern across multiple conditions including chronic fatigue syndrome, fibromyalgia, and metabolic disorders. Cellular energy production forms the foundation of resilient physiology.
Nervous System Regulation and Inflammatory Signaling
Another emerging area of research involves the role of autonomic nervous system regulation in chronic disease progression.
The autonomic nervous system controls heart rate, digestion, immune signaling, and inflammatory balance. It operates through two major branches: the sympathetic system responsible for stress responses and the parasympathetic system responsible for recovery and repair.
When sympathetic activation becomes chronic, inflammatory signaling tends to increase. Blood flow patterns change, digestion becomes impaired, and immune balance begins to shift.
This relationship between stress physiology and metabolic regulation is explored in the iThrive Academy article “The Nervous System and Obesity: Why Stress Physiology Drives Weight Gain.”
Although that article discusses metabolic disorders, the underlying principle applies equally to neurological disease.
The nervous system shapes immune behavior.
If chronic stress signals dominate the internal environment, inflammatory pathways become more active. In susceptible individuals, this inflammatory terrain may contribute to the development or progression of autoimmune conditions.
Environmental Triggers and Modern Lifestyle Stressors
Practitioners working with complex chronic diseases frequently observe the cumulative effects of environmental exposures.
These may include dietary patterns rich in inflammatory seed oils, chronic sleep deprivation, persistent psychological stress, exposure to environmental toxins, and nutrient deficiencies.
Over time, these stressors create a metabolic environment characterized by oxidative stress, mitochondrial dysfunction, and immune dysregulation.
Within this terrain, autoimmune responses become more likely.
The important insight for practitioners is that MS disease does not appear overnight. Instead, it develops gradually through interactions between genetic susceptibility and environmental pressures.
Understanding these upstream drivers allows clinicians to design more comprehensive strategies that address systemic health rather than focusing exclusively on symptom suppression.
Why Practitioners Must Think in Systems
If you are a practitioner, you may already sense this shift occurring within modern clinical thinking.
Traditional disease models divide the body into separate specialties. Neurology treats the brain. Gastroenterology treats the gut. Endocrinology treats hormones.
But chronic diseases rarely respect these boundaries.
Multiple sclerosis provides a powerful example of this limitation. The ms medical condition involves immune cells, neural tissue, gut microbiota, metabolic pathways, and mitochondrial energy production simultaneously.
A systems biology perspective allows practitioners to see these connections clearly.
This is precisely the framework emphasized within the iThrive Certified Functional Nutrition program, where practitioners learn to evaluate chronic diseases through interconnected physiological systems rather than isolated symptoms.
Students are trained to interpret lab markers, understand gut brain signaling, evaluate mitochondrial function, and apply nutrition based strategies that support cellular health.
In clinical practice, this knowledge becomes invaluable when working with individuals experiencing complex conditions like MS.
Clinical Implications for Practitioners
For practitioners seeking to support individuals living with multiple sclerosis symptoms, the most valuable shift may be conceptual rather than procedural.
Instead of viewing MS solely as an autoimmune attack, clinicians can begin evaluating the broader biological terrain.
This includes assessing gut barrier integrity, mitochondrial efficiency, nutrient status, inflammatory markers, sleep quality, stress physiology, and environmental exposures.
When these upstream drivers are addressed, the internal environment supporting neurological health may improve.
This systems oriented approach does not replace conventional treatments. Rather, it complements them by addressing underlying physiological imbalances that may influence disease progression.
Key Takeaway
Multiple sclerosis disease has long been described as a purely autoimmune condition. While immune mediated myelin damage is certainly central to the disease process, emerging research suggests that this explanation captures only part of the picture. The development of MS appears to involve interactions between immune signaling, mitochondrial energy production, gut microbiome balance, and nervous system regulation. For practitioners, this broader perspective offers an opportunity to rethink how chronic neurological disorders are understood and approached. By examining the systemic conditions that shape immune behavior and neural resilience, clinicians may move beyond symptom suppression toward a deeper understanding of biological regulation and long term health.










.jpg)











