Introduction
When most practitioners first study rheumatoid arthritis, the condition is introduced as a classic autoimmune disease. The immune system mistakenly targets synovial tissues within the joints, inflammation develops, and gradual joint damage follows. For decades, this explanation has shaped how the disease is taught in medical classrooms and how it is managed in clinical settings.
However, if you have ever worked closely with individuals living with rheumatoid arthritis symptoms, you may have noticed that the autoimmune description only tells part of the story. Patients rarely arrive in the clinic with joint inflammation appearing suddenly and in isolation. Instead, they often describe years of metabolic imbalance, persistent fatigue, digestive disturbances, chronic stress exposure, or weight regulation difficulties before joint pain becomes severe enough to seek medical help.
This observation raises an important question for practitioners and learners. If autoimmune arthritis is only the visible stage of the disease, what physiological changes are occurring long before joint damage appears?
Functional medicine and functional nutrition frameworks encourage practitioners to ask exactly this question. Instead of focusing solely on the immune attack within the joints, the functional perspective evaluates the systemic environment that allows inflammatory signaling to persist. Factors such as metabolic dysfunction, gut barrier disruption, nervous system stress responses, and nutrient deficiencies can all shape immune behavior.
For practitioners who want to deepen their understanding of chronic disease, this broader perspective is essential. Rheumatoid arthritis becomes less about isolated joint inflammation and more about understanding how the body’s regulatory systems interact.
Rheumatoid Arthritis as a Systemic Inflammatory Condition
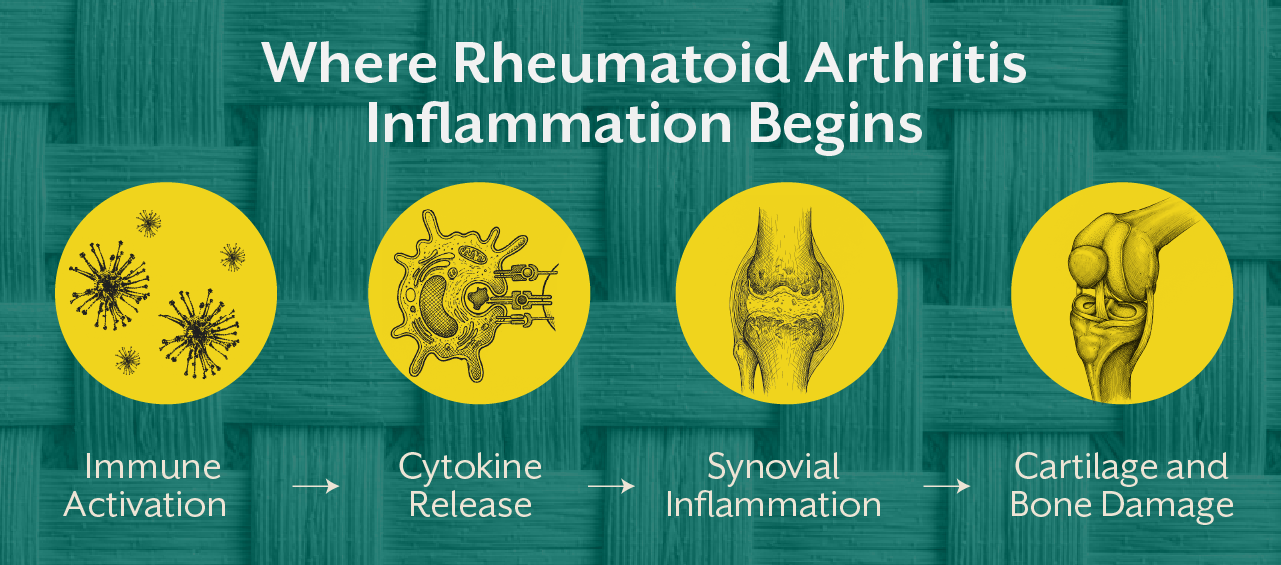
Although joint pain is the most recognizable symptom, inflammation in rheumatoid arthritis affects far more than the musculoskeletal system. The immune processes involved in the disease influence blood vessels, metabolic pathways, hormonal signaling, and nervous system regulation.
The synovial tissue lining the joints becomes infiltrated by immune cells such as macrophages and T cells. These cells release inflammatory cytokines that trigger swelling and tissue degradation. Over time, cartilage and bone structures begin to deteriorate.
Yet inflammation rarely originates exclusively inside the joint capsule. Instead, the inflammatory environment is shaped by systemic signals circulating throughout the body. Metabolic dysfunction, chronic stress, and immune activation in other organs can all contribute to this inflammatory terrain.
For example, patients experiencing autoimmune arthritis often show elevated markers of systemic inflammation such as C reactive protein and inflammatory cytokines long before severe joint damage appears. This indicates that the immune system is responding to broader physiological stressors.
Understanding rheumatoid arthritis through this systemic lens allows practitioners to ask more meaningful clinical questions. What metabolic signals are influencing immune activation? What environmental triggers may be sustaining inflammation? And perhaps most importantly, what biological systems should be evaluated beyond the joints?
The Gut Immune Connection in Autoimmune Arthritis
One of the most fascinating areas of research in rheumatoid arthritis functional medicine involves the relationship between gut health and immune regulation. The gastrointestinal tract is home to trillions of microorganisms that constantly interact with the immune system. These microbes produce metabolites that influence inflammation, immune tolerance, and even nervous system signaling.
When the balance of the gut microbiome becomes disrupted, a condition known as dysbiosis can develop. Dysbiosis often leads to increased intestinal permeability, sometimes described as a compromised gut barrier. When this barrier becomes weakened, bacterial fragments and inflammatory molecules can enter the bloodstream.
Once circulating in the body, these molecules stimulate immune activity and increase inflammatory signaling. Over time, this persistent immune stimulation may contribute to autoimmune responses in genetically susceptible individuals.
Researchers studying causes of rheumatoid arthritis have increasingly identified microbial patterns that differ between healthy individuals and those living with autoimmune arthritis. These findings reinforce the idea that immune dysregulation may begin far away from the joints.
This connection between gut health and systemic inflammation is explored in detail in the iThrive Academy blog “5 Pillars of Gut Brain Health: From Inflammation to Neurotransmitters.” That article highlights how microbial ecosystems influence immune signaling throughout the body, a concept highly relevant for practitioners studying chronic inflammatory diseases.

Cellular Energy and Inflammation in Rheumatoid Arthritis
Another dimension rarely discussed in conventional explanations of rheumatoid arthritis involves cellular energy metabolism. Every immune response requires energy. Immune cells must proliferate, migrate toward inflammatory sites, and produce signaling molecules. These processes depend heavily on mitochondrial function.
When mitochondrial efficiency declines, metabolic stress develops. Cells generate increased oxidative stress, inflammatory signaling becomes amplified, and tissue repair mechanisms weaken.
Emerging research suggests that individuals with autoimmune diseases often display signs of mitochondrial dysfunction. This includes reduced ATP production, elevated reactive oxygen species, and altered metabolic pathways within immune cells.
From a functional nutrition perspective, this connection is particularly important. Nutrient availability strongly influences mitochondrial health. Micronutrients such as magnesium, B vitamins, and antioxidants play essential roles in cellular energy metabolism.
If these nutrients are insufficient, cellular resilience declines and inflammatory processes may become more pronounced. This is one reason why nutrition for rheumatoid arthritis extends beyond simple anti-inflammatory diets. Supporting mitochondrial function may be a crucial component of regulating immune activity.
Practitioners exploring chronic disease mechanisms often see similar patterns across multiple conditions including chronic fatigue syndrome and fibromyalgia. Energy metabolism is deeply connected to immune regulation.
Nervous System Regulation and Chronic Inflammation
Another piece of the rheumatoid arthritis puzzle involves the autonomic nervous system. This system regulates stress responses, digestion, immune activity, and inflammatory balance.
When the body experiences persistent stress signals, the sympathetic branch of the nervous system becomes dominant. This state is often associated with increased cortisol release, altered immune signaling, and heightened inflammatory responses.
Chronic sympathetic activation may therefore contribute to the inflammatory environment associated with autoimmune diseases. In clinical practice, many individuals with rheumatoid arthritis report prolonged exposure to stress, sleep disruption, or emotional strain before disease onset.
The connection between nervous system regulation and metabolic disease is explored in the iThrive Academy article “The Nervous System and Obesity: Why Stress Physiology Drives Weight Gain.” While that article focuses on metabolic disorders, the physiological principles apply equally to autoimmune conditions.
Stress physiology influences immune behavior. When recovery mechanisms remain suppressed for long periods, inflammatory pathways can become chronically activated.
Metabolism, Weight Regulation, and Immune Signaling
An interesting clinical observation often overlooked in autoimmune disease discussions involves metabolic health. Individuals experiencing metabolic dysfunction or obesity frequently show higher levels of systemic inflammation.
Adipose tissue is not simply a storage site for excess energy. It functions as an endocrine organ capable of releasing inflammatory cytokines and signaling molecules. When adipose tissue expands significantly, these inflammatory signals increase.
This means metabolic health can directly influence immune regulation. Chronic metabolic stress may therefore create conditions that amplify inflammation in rheumatoid arthritis.
For practitioners, this insight highlights the importance of evaluating metabolic markers alongside autoimmune indicators. Blood glucose regulation, insulin signaling, and body composition may all influence the inflammatory environment within the body.
Functional nutrition frameworks emphasize this interconnected view of physiology. Rather than treating metabolic disorders and autoimmune diseases as separate categories, they are understood as overlapping manifestations of systemic imbalance.
Why Functional Nutrition Matters in Autoimmune Disease
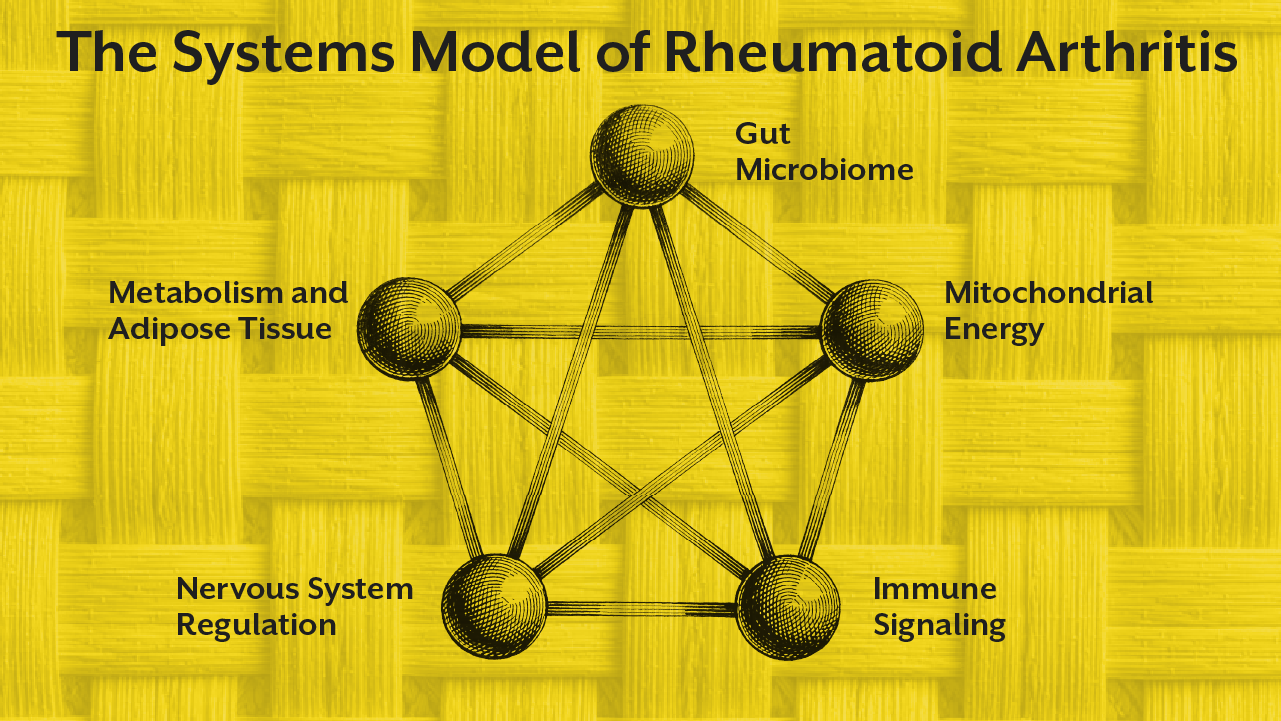
By now you may have noticed a recurring theme throughout this discussion. Rheumatoid arthritis is not simply a joint disorder. It represents the interaction of immune signaling, metabolic health, gut ecology, and nervous system regulation.
For practitioners, this realization opens an entirely different way of approaching chronic disease. Instead of focusing exclusively on symptom suppression, clinicians can begin exploring the upstream biological drivers that shape immune activity.
Functional nutrition plays a powerful role in this process. Nutritional strategies can influence gut microbiome diversity, mitochondrial energy production, inflammatory signaling, and hormonal balance.
This systems oriented approach forms the foundation of the iThrive Certified Functional Nutrition program, where practitioners learn to interpret laboratory markers, evaluate root causes of chronic disease, and design nutrition strategies that support long term metabolic resilience.
For many healthcare professionals, gaining this deeper systems understanding represents a major shift in how they approach clinical practice.
Why Functional Nutrition Matters in Autoimmune Disease
By now you may have noticed a recurring theme throughout this discussion. Rheumatoid arthritis is not simply a joint disorder. It represents the interaction of immune signaling, metabolic health, gut ecology, and nervous system regulation.
For practitioners, this realization opens an entirely different way of approaching chronic disease. Instead of focusing exclusively on symptom suppression, clinicians can begin exploring the upstream biological drivers that shape immune activity.
Functional nutrition plays a powerful role in this process. Nutritional strategies can influence gut microbiome diversity, mitochondrial energy production, inflammatory signaling, and hormonal balance.
This systems oriented approach forms the foundation of the iThrive Certified Functional Nutrition program, where practitioners learn to interpret laboratory markers, evaluate root causes of chronic disease, and design nutrition strategies that support long term metabolic resilience.
For many healthcare professionals, gaining this deeper systems understanding represents a major shift in how they approach clinical practice.
What Practitioners Often Miss in Rheumatoid Arthritis
Perhaps the most important lesson from the functional perspective is this. Autoimmunity does not arise in isolation. It emerges within a biological environment shaped by metabolism, stress physiology, microbiome dynamics, and nutrient status.
When practitioners evaluate these upstream systems, the clinical picture of rheumatoid arthritis becomes far more comprehensive. Instead of focusing solely on joint inflammation, clinicians begin to see the entire physiological network influencing immune behavior.
For practitioners and learners who want to expand their understanding of chronic disease, developing this systems thinking mindset is invaluable.
Key Takeaway
Rheumatoid arthritis has long been categorized as a purely autoimmune joint disease, yet emerging research and clinical observations suggest that this explanation captures only part of the biological reality. The inflammatory processes associated with rheumatoid arthritis appear to arise from complex interactions between immune signaling, metabolic health, gut microbiome balance, mitochondrial energy production, and nervous system regulation. For practitioners seeking a deeper understanding of chronic disease, this perspective is transformative. It shifts the focus away from isolated joint inflammation and toward the systemic environment that shapes immune behavior. Functional nutrition provides a framework for exploring these interactions and supporting the biological systems that influence long term health.










.jpg)











