Introduction
If you work clinically with thyroid disorders or teach others how to, you have likely experienced a recurring professional frustration. Patients present with persistent thyroid disease symptoms despite “normal thyroid levels.” Lab values appear stable. Medication has been prescribed. Yet fatigue continues, weight fluctuates, cognition slows, and metabolic resilience never fully returns.
At some point, you realise the limitation is not patient compliance, it is rather a model limitation.
Conventional thyroid education is largely gland-centric. It focuses on hormone production, replacement, and biochemical range normalisation. But thyroid physiology does not operate in isolation. Hormones are synthesised in one location, activated in another, transported through multiple regulatory systems, and ultimately expressed at the cellular level through metabolic signalling networks.
When one system destabilises, the entire thyroid regulatory architecture shifts.
This is where a systems biology framework becomes clinically essential. Instead of asking only how much hormone is produced, we begin asking:
- What determines hormone activation?
- What modifies cellular responsiveness?
- What regulates immune tolerance toward thyroid tissue?
- What governs metabolic energy required for hormone signalling?
- What environmental signals shape regulatory feedback?
From this perspective, thyroid dysfunction is not simply endocrine. It is ecological.
In this article, we will examine how gut health, mitochondrial function, and immune regulation integrate to shape thyroid physiology and why understanding this integration is essential for interpreting thyroid causes, managing Hashimoto’s thyroiditis, and designing meaningful thyroid treatment strategies.
Section 1 - Why Thyroid Physiology Is a Network, Not a Gland
The thyroid gland produces hormone. But hormone production alone does not define thyroid hormone function.
Consider what must happen after hormone release:
- The liver converts T4 into biologically active T3.
- Cells must generate sufficient ATP to respond to hormonal signals.
- Immune regulation must maintain tolerance to thyroid tissue.
- The nervous system modulates metabolic rate and signalling sensitivity.
- The gut microbiome influences inflammation, nutrient absorption, and hormone metabolism.
- This is not a linear process. It is a regulatory network.
Patients with Hashimoto’s thyroiditis illustrate this clearly. Thyroid antibodies do not emerge spontaneously. They arise from immune dysregulation influenced by intestinal permeability, antigen exposure, inflammatory signalling, and loss of immune tolerance.
Similarly, patients with persistent fatigue despite treatment often show impaired mitochondrial function, further limiting energy availability required for thyroid hormone signalling.
In systems biology, thyroid dysfunction is not viewed as a single failure point. It is viewed as a network adaptation.
Section 2 - The Gut - Thyroid Axis: Where Immune Tolerance Begins
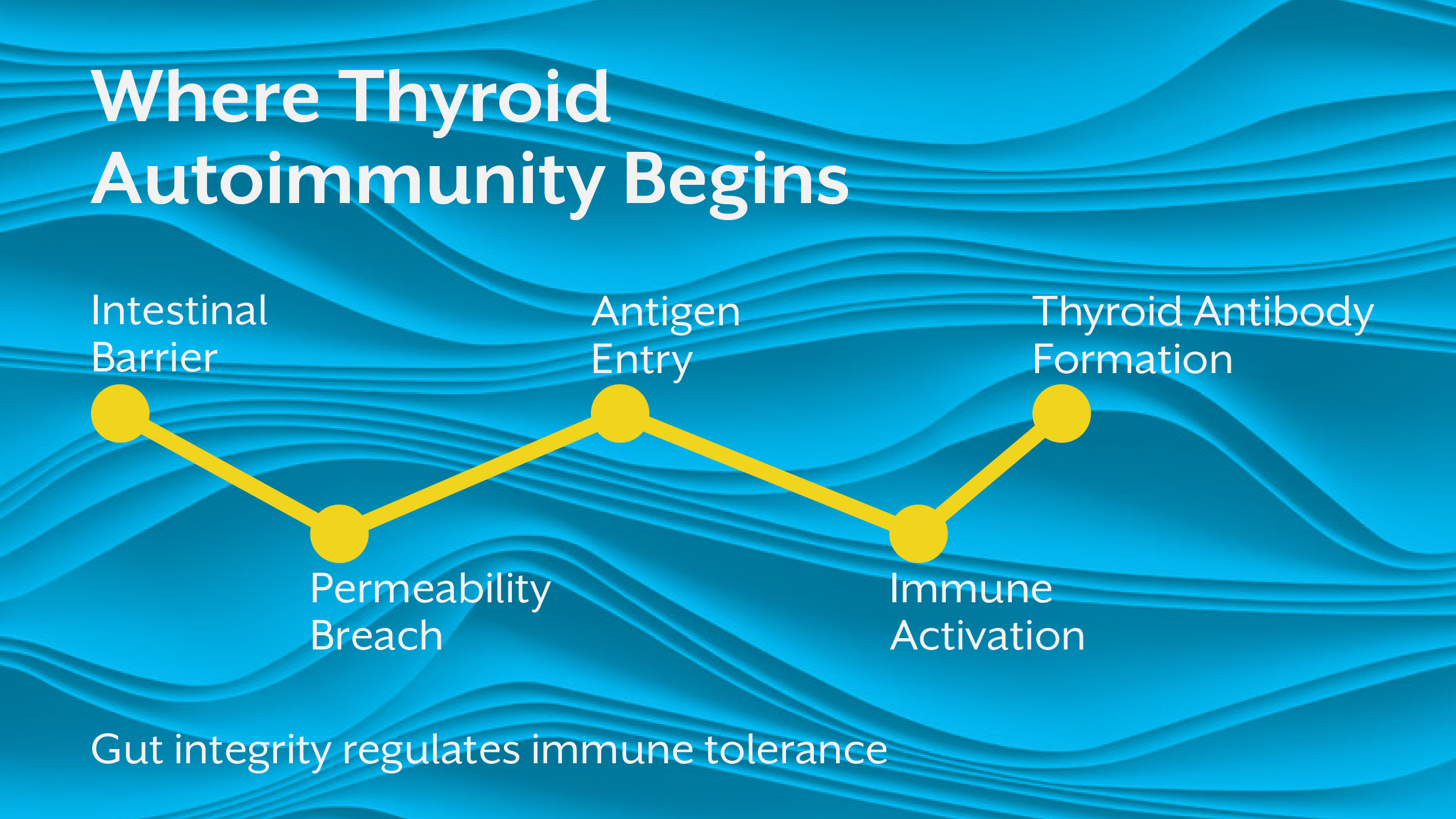
A large proportion of immune activity is regulated in the gut. This makes intestinal integrity central to thyroid autoimmunity.
When gut barrier function is compromised, antigenic proteins and microbial metabolites enter circulation. These activate immune pathways that may cross-react with thyroid tissue. Over time, this contributes to thyroid antibodies, a defining feature of Hashimoto’s thyroiditis.
But immune activation is only part of the story.
The gut also regulates:
- Selenium and zinc absorption is quite critical for thyroid hormone conversion
- Microbial deiodinase activity has an influence on hormone metabolism
- Inflammatory signalling that alters hypothalamic - pituitary regulation
This means that the gut shapes both immune activity and metabolic signalling, two central drivers of thyroid causes.
Clinically, this explains why patients with chronic digestive symptoms often experience thyroid disease symptoms that persist even when medication normalises lab values.
The gland may be treated. The regulatory environment is not.
Section 3 - Mitochondria: The Energy Infrastructure of Thyroid Signalling
Thyroid hormones regulate metabolic rate but metabolism cannot occur without cellular energy infrastructure.
Mitochondria determine how effectively cells respond to hormonal signalling. Even with adequate hormone availability, low mitochondrial efficiency reduces tissue responsiveness.
This produces a clinical paradox frequently seen in practice:
- Normal thyroid levels
- Persistent fatigue
- Cold intolerance
- Reduced metabolic flexibility
These are not always hormone production problems. They are energy utilisation problems.
Mitochondrial dysfunction also amplifies oxidative stress, which promotes immune activation and accelerates tissue damage further complicating Hashimoto’s thyroiditis treatment.
In other words, mitochondria influence both metabolic response and immune stability.
When evaluating thyroid hormone function, energy biology cannot be ignored.

Section 4 - Immune Regulation and Thyroid Tissue Vulnerability
Autoimmune thyroid conditions do not arise from random immune error. They emerge from loss of immune tolerance, a highly regulated process influenced by environmental triggers, inflammatory burden, and metabolic stress.
Immune cells require energy to maintain regulatory balance. Mitochondrial dysfunction therefore alters immune behaviour. At the same time, gut permeability increases antigen exposure, further stimulating immune activation.
This convergence explains why thyroid antibodies often rise during periods of systemic stress such as infection, chronic inflammation, or prolonged psychological load.
The immune system does not attack thyroid tissue in isolation. It responds to systemic signals.
Understanding this shifts the interpretation of thyroid disease symptoms from gland pathology to immune adaptation.
Section 5 - Nervous System Regulation of Thyroid Expression
One of the most under-recognised influences on thyroid physiology is autonomic nervous system regulation.
Chronic sympathetic activation alters metabolic priorities. The body shifts toward survival signalling, reducing energy allocation to growth, repair, and reproductive functions. Thyroid signalling is modulated accordingly.
Stress hormones alter hormone conversion patterns. Cellular receptor sensitivity shifts. Metabolic rate adapts downward.
This explains why patients under chronic stress may show worsening thyroid disease symptoms despite unchanged laboratory values.
The nervous system does not simply respond to thyroid function. It helps define how thyroid signals are expressed.
Section 6 - Why Reductionist Thyroid Treatment Often Falls Short
When thyroid treatment focuses only on hormone replacement, the broader regulatory environment remains unchanged.
If gut inflammation persists, immune activation continues.
If mitochondrial function is impaired, energy signalling remains limited.
If stress physiology remains dysregulated, conversion patterns remain altered.
This explains why some patients with normal thyroid levels still experience symptoms and why long-term management of Hashimoto’s thyroiditis requires multi-system intervention.
Clinical education that ignores regulatory integration leaves practitioners managing numbers rather than physiology.
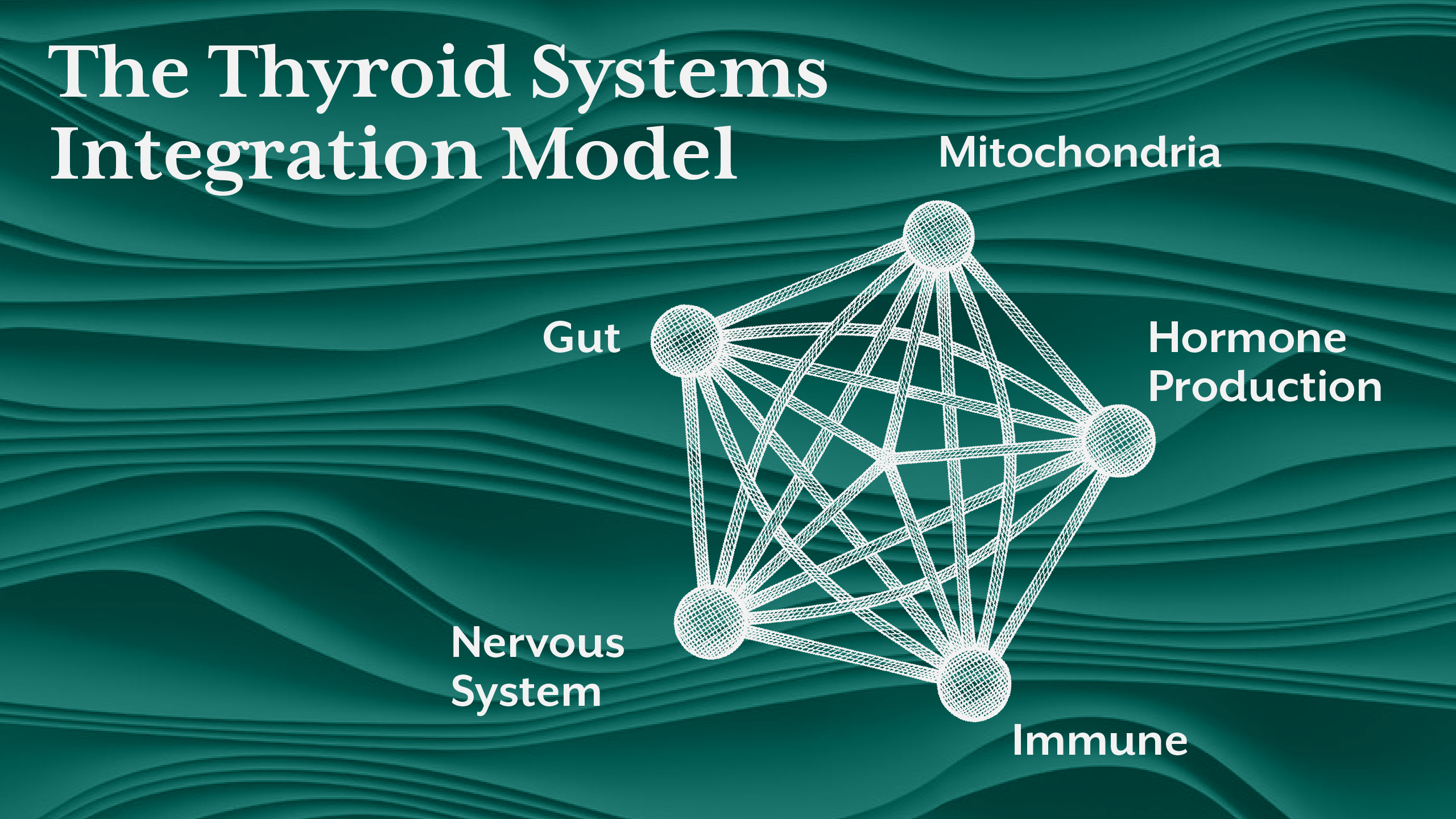
Section 7 - Building a Systems-Based Clinical Framework
A practitioner-level thyroid assessment must move beyond gland measurement toward regulatory mapping.
This includes evaluating:
- Immune activation patterns
- Gut integrity markers
- Mitochondrial function indicators
- Stress physiology and autonomic balance
- Cellular responsiveness indicators
This integrated framework is increasingly central to advanced clinical education including programs at iThrive Academy and the iThrive Certified Functional Nutrition (iCFN) clinical curriculum.
Practitioners trained in systems interpretation develop greater diagnostic precision because they assess drivers, and not just outputs.
Section 8 - Educational Implications for Practitioners
If you are teaching thyroid physiology or learning to interpret complex cases, the critical shift is conceptual.
You are no longer asking:
“What is the thyroid doing?”
You are asking:
“What is regulating thyroid expression across systems?”
This shift transforms clinical reasoning. It also changes how treatment strategies are prioritised, sequenced, and evaluated.
Many practitioners recognise this gap when patients fail to respond predictably to intervention. That moment of uncertainty often marks the transition from protocol-based care to systems-based interpretation.
Structured learning pathways such as advanced thyroid-focused training and specialised educator programs are designed to support this transition from biochemical management to physiological understanding.
Key Takeaway
A systems biology approach reframes thyroid disorders from isolated endocrine dysfunction to network-level physiological adaptation. Hormone production, immune tolerance, mitochondrial energy generation, gut integrity, and nervous system signalling form an interconnected regulatory web that determines how thyroid physiology is expressed in real patients. When practitioners evaluate thyroid disease symptoms through this integrated lens, previously unexplained clinical patterns begin to make sense in forms of persistent symptoms despite normal thyroid levels, fluctuating antibody activity, variable treatment response, and metabolic instability. The future of thyroid care lies not in refining single markers but in understanding how biological systems communicate, compensate, and adapt over time. Clinical mastery begins when we stop treating glands and start interpreting networks.









.jpg)











